The Uses and Dangers of Aluminum Chloride
The Uses and Dangers of Aluminum Chloride
Aluminum chloride is a chemical compound that is used primarily in making aluminum metal, but it has other uses, as well. The compound reacts strongly in the presence of water, so it’s important to take care when handling it.
Chemical Properties
Aluminum chloride is a white compound of aluminum and chloride. Iron trichloride can often contaminate samples of this compound, which may give it a yellow color. Its solid form has very low melting and boiling points. In its anhydrous (without water) form, aluminum chloride reacts strongly with both water and bases (they can bind with hydrogen), so it’s important to keep the compound away from substances that contain either of them. Even trace bits of moisture can cause a reaction with dry aluminum chloride. The chemical formula for aluminum chloride is AlCl₃.

Industrial Uses
The primary uses of aluminum chloride are in manufacturing and industry. First and foremost, it’s a component in the production of aluminum, in metallurgy, and as an ingredient in aluminum smelting. It’s also used in manufacturing petrochemicals like ethylbenzene and alkylbenzene. Certain kinds of pharmaceuticals require aluminum chloride as an ingredient. Its many other applications include the production of paint, synthetic rubber, lubricants, wood preservatives, and some organic chemicals. This is a versatile compound.
 In the Home
In the Home
A use that may come closer to home for most people is that there’s a tiny bit of aluminum chloride in many antiperspirants. In fact, larger amounts of it are in prescription antiperspirants. Aluminum chloride works in deodorant by combining with electrolytes in the skin to create a gel plug in the sweat glands. It also has a slightly astringent effect on the pores, causing them to contract, which keeps the pores from releasing sweat. Because of this ability to contract pores, it’s also an ingredient in cosmetic astringents.
Dangers of Using It
Aluminum chloride is potentially hazardous to humans, particularly in its anhydrous form. It’s highly corrosive and can lead to serious damage if it’s inhaled, ingested, or touches the skin. Inhalation can lead to severe irritation of the nose and throat, difficulty in breathing, headache, nausea, and vomiting. Severe eye irritation and the risk of permanent damage to the eyes can happen if it gets in the eyes. Skin contact can lead to extreme irritation or severe burns. Ingestion can result in permanent damage to the digestive system.
Preventing Exposure
Household forms of aluminum chloride are not harmful to humans. Exposure to aluminum chloride generally only happens in a laboratory or industrial setting. In these cases, the managers of these facilities must take precautions to make sure people aren’t exposed. These include respiratory protection and suitable ventilation, chemical resistant PVC gloves, chemical-proof goggles or face shields, and PVC aprons and boots when splashing is a risk. Management should offer decontamination showers and eyewash stations as a precaution.
Aluminum chloride is a compound of aluminum and chloride that’s widely used in petroleum refining and the manufacturing of many products. In addition, antiperspirants and cosmetic astringents use this compound. In its raw form, it may be hazardous to humans, so make sure you know how to protect yourself if you have to use it!
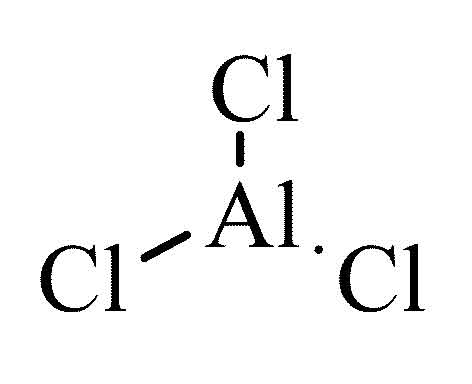 Formula for Aluminum Chloride
Formula for Aluminum Chloride
AlCl3
Properties for Aluminum Chloride
Molar mass: 133.34 g/mol (anhydrous), 241.43 g/mol (hexahydrate)
Melting Point: 192.4 °C (378.3 °F; 465.5 K) (anhydrous), 100 °C (212 °F; 373 K)(hexahydrate), 180 °C (356 °F; 453 K) (sublimes)
Boiling Point: 120 °C (248 °F; 393 K) (hexahydrate)
Density: 2.48 g/cm3 (anhydrous); 1.3 g/cm3 (hexahydrate)
 Alloy Suppliers
Alloy Suppliers Aluminum
Aluminum Aluminum Extrusions
Aluminum Extrusions Copper-Brass-Bronze
Copper-Brass-Bronze Magnets
Magnets Nickel
Nickel Stainless Steel
Stainless Steel Stainless Steel Tubing
Stainless Steel Tubing Steel Service Centers
Steel Service Centers Titanium
Titanium Tungsten
Tungsten Wire Rope
Wire Rope Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services