Aluminum carbonate is an aluminum salt mainly used in the medical field to treat stomach and intestinal problems. Some chemist argue that it doesn’t existence since it is such an unstable substance. To keep it from evaporating, it has to be placed under pressure and away from water.
Aluminum carbonate is an aluminum salt mainly used in the medical field to treat stomach and intestinal problems. Some chemist argue that it doesn’t existence since it is such an unstable substance. To keep it from evaporating, it has to be placed under pressure and away from water.
Uses
As a medicine, aluminum carbonate is used as an anti-acid for acid reflux, stomach inflammation, and ulcers. It can be ingested in pill or liquid form to control gastrointestinal disorders. Care has to be taken in the use of aluminum carbonate since it can decrease the effects of other medications.
Overuse of aluminum carbonate can lead to constipation and slight discomfort. Some people are allergic to aluminum products and should not take aluminum carbonate. Though there are over the counter varieties, it is best to consult a doctor regarding its use.
A necessary element for the body is phosphorus, which is used with calcium to build strong bones. If your body has too much or too little, you may contract either hyperphosphatemia or hypophosphatemia. Aluminum carbonate helps control the level of phosphorus or phosphates and prevents the formation of urinary tract stones.
How aluminum carbonate is formed
Aluminum carbonate is formed by the fusing of an aluminum base with an alkali carbon metal. Once it forms, it immediately decomposes because of hydrolysis in the aluminum carbonate molecule. The reason for its decomposition is the weak acid and its symbiosis with a poor base that cannot fight atmospheric conditions. The instability of aluminum carbonate make it impossible to store and study. Since it has no commercial value, and storing it would require very specialized conditions, little is done to create a laboratory version.
When aluminum carbonate decomposes, it becomes aluminum oxide and carbon dioxide gas. During the mixing process, the aluminum ion immediately separates from the oxygen atom, which combines with carbon to make carbon oxide. The chemical formula for aluminum carbonate is Al2(CO3)3.
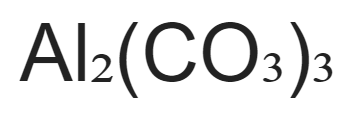
Aluminum Hydrogen Carbonate
Aluminum hydrogen carbonate, also known as aluminum bicarbonate, is closely related to aluminum carbonate. Much like aluminum carbonate, aluminum hydrogen carbonate does not exist except in formula form. The difficulty in producing it is the inability of hydrogen to bond with a metal. Aluminum hydrogen carbonate is a member of a group of inorganic salts.
 Alloy Suppliers
Alloy Suppliers Aluminum
Aluminum Aluminum Extrusions
Aluminum Extrusions Copper-Brass-Bronze
Copper-Brass-Bronze Magnets
Magnets Nickel
Nickel Stainless Steel
Stainless Steel Stainless Steel Tubing
Stainless Steel Tubing Steel Service Centers
Steel Service Centers Titanium
Titanium Tungsten
Tungsten Wire Rope
Wire Rope Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services